Ocean acidification
Definition of Ocean acidification:
The long-term decrease in seawater pH caused mainly by the uptake of anthropogenic carbon dioxide from the atmosphere. Dissolved CO2 alters seawater carbonate chemistry, increasing hydrogen ion activity and reducing carbonate ion availability.
This is the common definition for Ocean acidification, other definitions can be discussed in the article
|
This article introduces processes involved in ocean acidification and summarizes results from studies on the impact of ocean acidification on several common calcifying marine organisms. Ocean acidification is mainly caused by anthropogenic release of carbon dioxide into the atmosphere and subsequent uptake of CO2 by the ocean. The article first defines ocean acidity and alkalinity, and then explains the counterintuitive result that calcification can promote acidification, whereas dissolution of calcium carbonate counteracts acidification and can enhance the uptake of atmospheric [math]CO_2[/math].
Contents
- 1 Ocean acidity
- 2 Alkalinity
- 3 Carbonate chemistry
- 4 Feedbacks to acidification
- 5 Deep sea carbonate dissolution
- 6 Ecosystem impacts of ocean acidification
- 7 Influence of ocean acidification on a few bivalve species
- 8 Influence of ocean acidification on coccolithophores
- 9 Bioavailability of trace metals
- 10 Related articles
- 11 References
Ocean acidity
Ocean acidity is measured by [math]pH[/math], a measure of hydrogen ion activity[1]. Because ion activity cannot be measured directly, [math]pH[/math] is often approximated by
[math]pH \approx - \log_{10} ([H^+]) , \qquad (1)[/math]
where [math][H^+][/math] is the hydrogen ion concentration. In seawater, [math]pH[/math] depends on temperature, salinity, pressure and the pH scale used. In simple aqueous solutions, a solution is neutral if [math]pH=7[/math], acidic if [math]pH\lt 7[/math] and basic if [math]pH\gt 7[/math].
The average [math]pH[/math] of ocean surface waters was about 8.15 in pre-industrial times. Due to the increase in atmospheric [math]CO_2[/math], the amount of [math]CO_2[/math] dissolved in the ocean has also increased. It is estimated that about 30% of yearly emitted [math]CO_2[/math] is absorbed by the oceans[2]. This uptake has lowered the average surface-ocean [math]pH[/math] to about 8.05 in 2020, equivalent to an increase in hydrogen ion activity of about 26%.
Surface-ocean [math]pH[/math] is projected to decrease further by about 0.14–0.43 units by 2100, depending on the emissions scenario, with a concurrent increase in sea surface temperature of about 2–4 oC[3].
Ocean acidification is not caused only by atmospheric [math]CO_2[/math] uptake. Other processes can also contribute locally or regionally, including calcification, decomposition of organic material, nitrification in surface waters promoted by sewage discharge, and oxidation reactions in sediments[4]. Acidity is not directly related to dissolved oxygen concentration, because [math]O_2[/math] and [math]H^+[/math] do not react directly to form [math]H_2O[/math].
Alkalinity
Alkalinity plays a major role in ocean chemistry, [math]CO_2[/math] storage and calcium carbonate precipitation and dissolution. Alkalinity is defined as the excess of proton acceptors over proton donors in seawater[5]. Alkalinity and [math]pH[/math] are generally positively correlated, although the relationship also depends on dissolved inorganic carbon and other chemical constituents.
Total alkalinity (TA) is a measure of the capacity of seawater to neutralize added acid. It is largely controlled by bicarbonate and carbonate ions and is an indicator of the ocean's capacity to store [math]CO_2[/math]. Total alkalinity can be approximated by[6]:
[math]TA = [HCO_3^-] + 2[CO_3^{2-}] + [OH^-] - [H^+] + \mathrm{minor \, components} .[/math]
Alkalinity is commonly determined by measuring the quantity of strong acid required to titrate seawater to a defined endpoint[7].
Natural weathering of rocks is the primary long-term source of ocean alkalinity. For example, dissolution of olivine can be represented schematically as
[math]Mg_2SiO_4 + 4CO_2 + 4H_2O \rightarrow 2Mg^{2+} + 4HCO_3^- + H_4SiO_4 .[/math]
This reaction increases alkalinity and converts dissolved [math]CO_2[/math] mainly into bicarbonate ions[8].
Oxidation reactions involving oxygen, such as aerobic mineralization, generally consume alkalinity. In contrast, anaerobic processes often produce alkalinity. For example, during denitrification, nitrate is reduced to dinitrogen gas and alkalinity is generated. Similarly, burial of reduced sulfur in marine sediments contributes to alkalinity generation over longer timescales. Alkalinity is also generally generated by mineral dissolution[5].
Carbonate chemistry
The uptake of atmospheric [math]CO_2[/math] increases dissolved inorganic carbon (DIC) and alters seawater carbonate chemistry according to the equilibria[9]
[math]CO_2 + H_2O \rightleftharpoons H_2CO_3 \rightleftharpoons HCO_3^- + H^+ \rightleftharpoons CO_3^{2-} + 2H^+ . \qquad (2)[/math]
At typical surface-ocean [math]pH[/math] near 8.1, DIC consists mainly of bicarbonate [math]HCO_3^-[/math], with a smaller fraction as carbonate [math]CO_3^{2-}[/math] and only a small fraction as dissolved [math]CO_2[/math] and carbonic acid. Typical proportions are about 90% bicarbonate, about 9% carbonate and less than 1% dissolved [math]CO_2[/math] plus carbonic acid, although these fractions depend on temperature, salinity, pressure, alkalinity and [math]pH[/math].
On geological timescales, mineral weathering of silicate and carbonate rocks supplies much of the alkalinity and DIC to the ocean[6]. On anthropogenic timescales, however, uptake of atmospheric [math]CO_2[/math] directly increases DIC and shifts carbonate equilibria.
Uptake of [math]CO_2[/math] leads to ocean acidification because it increases hydrogen ion activity. Much of the added [math]CO_2[/math] reacts with carbonate ions, giving[10]
[math]CO_2 + H_2O + CO_3^{2-} \rightleftharpoons 2HCO_3^- . \qquad (3)[/math]
This reaction shows that uptake of [math]CO_2[/math]:
- reduces the concentration of carbonate ions [math]CO_3^{2-}[/math], making less carbonate available for marine calcifying organisms that produce calcium carbonate [math]CaCO_3[/math] shells or skeletons;
- increases the fraction of DIC present as bicarbonate, thereby buffering the rise in dissolved [math]CO_2[/math] and enhancing the ocean's capacity to store carbon.
With increasing [math]pH[/math], a larger fraction of DIC occurs as carbonate [math]CO_3^{2-}[/math], increasing the buffering capacity of seawater. With decreasing [math]pH[/math], a smaller fraction of DIC occurs as carbonate, reducing the ocean's capacity to absorb additional [math]CO_2[/math] without a large rise in dissolved [math]CO_2[/math].
Feedbacks to acidification
The presence of calcium carbonate [math]CaCO_3[/math] in the ocean provides an important feedback mechanism that mitigates ocean acidification. Dissolution of calcium carbonate in seawater can proceed through several pathways[11]:
[math]CaCO_3 \rightleftharpoons Ca^{2+} + CO_3^{2-}; \quad CaCO_3 + H_2O \rightleftharpoons Ca^{2+} + HCO_3^- + OH^-; \quad CaCO_3 + H_2O + CO_2 \rightleftharpoons Ca^{2+} + 2HCO_3^- . \qquad (4)[/math]
The carbonate and bicarbonate ions produced by dissolution can consume hydrogen ions or buffer their increase. Dissolution of calcium carbonate therefore counteracts acidification caused by [math]CO_2[/math] uptake[12]. Increased alkalinity in surface waters lowers dissolved [math]CO_2[/math] for a given DIC concentration and can increase the capacity of seawater to absorb atmospheric [math]CO_2[/math].
Conversely, calcification by marine organisms consumes carbonate ions and alkalinity. Precipitation of calcium carbonate can be represented by the reverse of the last reaction in Eq. (4):
[math]Ca^{2+} + 2HCO_3^- \rightarrow CaCO_3 + CO_2 + H_2O .[/math]
Thus, production of 1 mole of [math]CaCO_3[/math] consumes 2 moles of bicarbonate and can release [math]CO_2[/math] locally. This tends to reduce ocean uptake of atmospheric [math]CO_2[/math], although the amount of [math]CO_2[/math] released is partly buffered by seawater chemistry[6].
Deep sea carbonate dissolution
Ocean surface waters are typically supersaturated with respect to calcium carbonate due to high concentrations of calcium and carbonate ions. Despite supersaturation with respect to calcite and aragonite, inorganic precipitation is limited because crystal growth is inhibited by other ions, including [math]Mg^{2+}[/math][13]. Many marine organisms have evolved biological mechanisms to precipitate [math]CaCO_3[/math] as calcite or aragonite.
The solubility of calcium carbonate increases as the carbonate saturation state decreases. With increasing depth, lower temperature, higher pressure, accumulation of respired [math]CO_2[/math] and lower carbonate ion concentrations generally increase the solubility of [math]CaCO_3[/math]. This explains why carbonate particles tend to dissolve at depth. Dissolution of [math]CaCO_3[/math] in the deep ocean raises alkalinity and helps buffer changes in seawater [math]pH[/math][11].
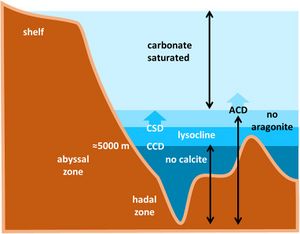
Calcium carbonate occurs in the ocean mainly in two crystalline forms: aragonite and calcite. The lysocline is the depth interval over which calcium carbonate dissolution increases rapidly. Its upper limit is related to the calcite saturation depth (CSD), and its lower limit approaches the calcite compensation depth (CCD), below which carbonate accumulation in sediments is strongly reduced or absent. Below the CCD, seafloor sediments contain little or no carbonate minerals, forming the so-called carbonate snow line.
Aragonite is more soluble than calcite. The aragonite saturation horizon and aragonite compensation depth are therefore shallower than their calcite equivalents. Organisms that produce aragonite are generally more vulnerable to acidification than organisms that produce calcite. Dissolution of aragonite in the deep sea releases alkalinity and can raise the [math]CaCO_3[/math] saturation state, thereby helping to protect calcite deposits from dissolution[15].
Global ocean modeling suggests that the CCD has already risen by nearly 100 m on average since pre-industrial times and may rise by several hundred meters more this century. As a result, potentially millions of square kilometres of ocean floor may undergo a rapid transition in overlying water chemistry, with calcareous sediment becoming unstable and the carbonate snow line rising[14].
Ecosystem impacts of ocean acidification
Although ocean acidification is affecting marine ecosystems worldwide, it is difficult to attribute observed ecosystem changes unambiguously to acidification alone. Ecosystem changes may also result from natural variability, global warming, deoxygenation, eutrophication, fishing and other human impacts. Long-term biological and ecological measurements needed to distinguish trends from natural variability remain scarce. Studies of organism responses are often based on controlled experiments, but these provide only limited insight into population- and ecosystem-level impacts because acidification effects can cascade through communities in complex ways[16].
Field-based observations of acidification impacts often use natural [math]CO_2[/math] gradients. Agostini et al. (2018[17]) compared intertidal and subtidal rocky reef communities near shallow volcanic seeps along natural gradients in [math]CO_2[/math]. These gradients ranged from present-day values to projected future values and were associated with decreasing aragonite saturation and increasing dissolved inorganic carbon. Abrupt changes in intertidal and subtidal rocky reef communities were observed, including biodiversity loss, decline of habitat-forming species such as coralline algae, canopy-forming macroalgae, scleractinian corals and barnacles, and an increase in low-profile turf algae. A related transplant study near the same seeps showed that algal communities exposed to [math]CO_2[/math]-enriched waters became dominated by turf algae with lower biomass, diversity and structural complexity, a pattern consistent across seasons[18]. Communities partly recovered after being transplanted back to non-enriched conditions.
These observations suggest that acidification can shift ecosystems in subtropical-temperate transition zones from complex calcified biogenic habitats towards less complex non-calcified habitats. Acidification may also impede the poleward expansion of some coral communities under warming conditions[17]. A comprehensive open-access review of possible impacts of ocean acidification on marine benthic ecosystems is provided by Somma et al. (2023[19]).
Another possible impact of ocean acidification is a decrease in algal production of dimethyl sulfide (DMS), which could influence cloud formation and ocean albedo; see Greenhouse gas regulation.
Influence of ocean acidification on a few bivalve species
A literature review[20] indicates that many bivalves reduce metabolic activity under low [math]pH[/math]. However, bivalves from regions with naturally lower [math]pH[/math], such as upwelling systems, may show neutral or positive responses, suggesting local adaptation or acclimatization. Bivalves in warmer regions may be more sensitive to low [math]pH[/math].
Oyster Magallana gigas and mussel Mytilus spp.
Mytilus spp. and Magallana gigas together account for almost half of global mollusc production in aquaculture. The effects of acidification depend partly on shell structure and composition. Both Mytilus spp. and Magallana gigas form calcite layers, while Mytilus spp. also form an inner aragonite layer. Aragonite is more soluble than calcite and is therefore generally more vulnerable to ocean acidification.
Mele et al. (2023[21]) examined the interactive effects of [math]pH[/math] (8.1 versus 7.7), temperature (12 versus 14 oC) and feeding (control versus extra feed) on Mytilus spp. and Magallana gigas in a full factorial experiment. They reported that warming and low [math]pH[/math] altered biomineralization pathways in Mytilus spp., but shell growth, thickness and hardness were partly maintained. Under low [math]pH[/math], Mytilus spp. increased environmentally sourced carbon in aragonite, while M. gigas showed changes in nutrient-related indicators but maintained shell growth and biomineralization pathways.
Previous research has shown that increased food supply can reduce shell corrosion and support shell growth in molluscs exposed to acidification. In the Mele et al. study, M. gigas showed overall greater shell performance and resilience than Mytilus spp.
White furrow shell Abra alba
Vlaminck et al. (2022[22]) studied the physiological response of the white furrow shell Abra alba to three [math]pH[/math] treatments: [math]pH = 8.2[/math], [math]pH = 7.9[/math] and [math]pH = 7.7[/math]. They found no effect of [math]pH[/math] on survival.
However, at [math]pH \sim 7.7[/math], respiration and calcification rates decreased, energy intake was reduced and metabolic losses increased. These responses resulted in negative scope for growth and a lower condition index, suggesting that short-term survival may not imply long-term physiological resilience or maintained ecosystem functioning.
Peruvian scallop Argopecten purpuratus
Along the Peruvian coast, the Peruvian scallop Argopecten purpuratus naturally experiences low [math]pH[/math] conditions of about 7.6–8.0 due to coastal upwelling. Cordova-Rodríguez et al. (2022[23]) exposed juvenile scallops for 28 days to unmanipulated seawater with [math]pH = 7.8[/math] and to a low-pH treatment of [math]pH = 7.4[/math].
At the end of the experiment, shell height, shell weight, growth rate and calcification rate were reduced under low [math]pH[/math]. Shell microhardness increased, while crushing force and soft tissue mass did not differ significantly between treatments. These results suggest that low [math]pH[/math] can reduce shell growth while altering shell mechanical properties in ways that may provide partial protection.
Influence of ocean acidification on coccolithophores
Coccolithophores are unicellular phytoplankton covered with calcium carbonate plates called coccoliths. The most common species is Emiliania huxleyi, which occurs widely in temperate, subtropical and tropical oceans. Coccoliths from dead coccolithophores contribute to ocean's carbon sink, but calcification itself is not equivalent to atmospheric [math]CO_2[/math] sequestration, because calcium carbonate formation releases [math]CO_2[/math]. Dead coccolithophores sink slowly, and much of their material may dissolve or be remineralized before reaching the seafloor. Coccoliths and coccospheres are more likely to be transported to depth when incorporated into fecal pellets or marine snow[24].
A major fraction of pelagic calcium carbonate production in surface waters is associated with coccolithophores, but their contribution to the [math]CaCO_3[/math] stock buried in deep-sea sediments may be comparable to, or not much larger than, that of less abundant but larger calcifiers such as foraminifera[25].
Krumhardt et al. (2019[26]) studied the sensitivity of coccolithophore growth and calcification to increasing [math]CO_2[/math] using the Community Earth System Model version 2.0[27]. The model was evaluated against satellite-derived particulate inorganic carbon, shipboard estimates of coccolithophore biomass, compilations of calcification rates and estimates of globally integrated upper-ocean calcification.
The model results indicate that increasing [math]CO_2[/math] can stimulate coccolithophore growth in some regions, including parts of the North Atlantic, western Pacific and Southern Ocean. However, elevated [math]CO_2[/math] generally impairs calcification. Most ocean regions show large declines in pelagic calcification under high end-of-century [math]CO_2[/math] levels, and coccolithophores are projected to be more lightly calcified overall.
The findings of Ziveri et al. (2023)[25] suggest that calcium carbonate production by coccolithophores and carbon export to the deep sea are not strongly coupled. Export depends on several processes, including grazing, particle aggregation, organic-to-inorganic carbon ratios in aggregates and the relative abundance of foraminifera, coccolithophores and pteropods. A decrease in calcification by coccolithophores can provide a negative feedback to acidification because less alkalinity is exported from surface waters, allowing additional atmospheric [math]CO_2[/math] uptake.
Bioavailability of trace metals
Trace metals such as iron, nickel, copper, zinc and cadmium are essential for microorganisms that support ocean primary production. If trace metal concentrations are too low, biological production can be limited; if they are too high, metals can become toxic. This balance is delicate because marine organisms are adapted to existing trace-metal availability.
For any substance, including metals, nutrients and contaminants, the fraction that can be readily used by organisms is called bioavailable. The bioavailability of metals is influenced by chemical speciation. Many metals form complexes with hydroxide and carbonate ions. Under acidified conditions, concentrations of hydroxide [math][OH^-][/math] and carbonate [math][CO_3^{2-}][/math] decrease, potentially increasing the concentration of some bioavailable metal forms, including forms of Fe and Cu[28]. A modest increase in Fe availability could stimulate primary production in iron-limited regions, whereas increased Cu bioavailability may have toxic effects.
However, this is not the whole story. A large fraction of many trace metals is bound to organic ligands, a process known as chelation. Acidic sugars, exopolysaccharides and other exopolymeric substances in seawater can bind metal ions through phenolic, carboxylic and other functional groups[29]. How these compounds and their metal-binding properties respond to increasing acidity is not yet fully understood. Environmental factors such as sunlight and reactive oxygen species can also dissociate metals from organic complexes through redox reactions or ligand degradation. Several biogeochemical models have been developed to simulate the impact of acidification on trace-metal bioavailability, but clear predictions remain difficult because seawater contains many organic compounds with poorly known chemistry and metal-binding affinities[30][31].
Related articles
- Ocean carbon sink
- Effects of global climate change on European marine biodiversity
- Greenhouse gas regulation
- Blue carbon sequestration
See also Wikipedia: Ocean acidification.
References
- ↑ Marion, G.M., Millero, F.J., Camoes, M.F., Spitzer, P., Feistel, R. and Chen, C.T.A. 2011. pH of seawater. Marine Chemistry 126: 89-96
- ↑ Terhaar, J., Froelicher, T.L. and Joos, F. 2022. Observation-constrained estimates of the global ocean carbon sink from Earth system models. Biogeosciences 19: 4431–4457
- ↑ Lee, J.-Y., Marotzke, J., Bala, G., Cao, L., Corti, S., Dunne, J.P., Engelbrecht, F., Fischer, E., Fyfe, J.C., Jones, C., Maycock, A., Mutemi, J., Ndiaye, O., Panickal, S. and Zhou, T. 2021. Chapter 4: Future Global Climate: Scenario-Based Projections and Near-Term Information. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, pp. 553–672
- ↑ Wallace, R.B. and Gobler, C.J. 2021. The role of algal blooms and community respiration in controlling the temporal and spatial dynamics of hypoxia and acidification in eutrophic estuaries. Marine Pollution Bulletin 172, 12908
- ↑ 5.0 5.1 Middelburg, J. J., Soetaert, K. and Hagens, M. 2020. Ocean alkalinity, buffering and biogeochemical processes. Reviews of Geophysics 58, e2019RG000681
- ↑ 6.0 6.1 6.2 Pernet, F., Dupont, S., Gattuso, J-P., Metian, M. and Gazeau, F. 2024. Cracking the myth: Bivalve farming is not a CO2 sink. Rev Aquac. 2024: 1–13
- ↑ Millero, F.J., Zhang, J.Z., Lee, K. and Campbell, D.M. 1993. Titration alkalinity of seawater. Mar. Chem. 44: 153–165
- ↑ Renforth, P. and Henderson, G. 2017. Assessing ocean alkalinity for carbon sequestration. Rev. Geophys. 55: 636–674
- ↑ Mehrbach, C., Culberson, C.H., Hawley, J.E. and Pytkowicz, R.M. 1973. Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnol. Oceanogr. 18: 897–907
- ↑ Egleston, E.S., Sabine, C.L. and François, M.M.M. 2010. Revelle revisited: Buffer factors that quantify the response of ocean chemistry to changes in DIC and alkalinity. Global Biogeochemical Cycles 24, GB1002
- ↑ 11.0 11.1 Batchelor-McAuley, C., Yang, M., Rickaby, R.E.M. and Compton, R.G. 2022. Calcium Carbonate Dissolution from the Laboratory to the Ocean: Kinetics and Mechanism. Chem.Eur.J. 28, e2022022
- ↑ Zeebe, R. E. and Wolf‐Gladrow, D. 2001. CO2 in seawater: Equilibrium, kinetics, isotopes. In Elsevier Oceanography Series (360 pp.)
- ↑ Berner, R. A. 1975. The role of magnesium in the crystal growth of calcite and aragonite from sea water. Geochim. Cosmochim. Acta 39: 489–504
- ↑ 14.0 14.1 Harris, P.T., Westerveld, L., Zhao, Q. and Costello, M.J. 2023. Rising snow line: Ocean acidification and the submergence of seafloor geomorphic features beneath a rising carbonate compensation depth. Marine Geology 463, 107121
- ↑ Sulpis, O., Agrawal, P., Wolthers, M., Munhoven, G., Walker, M. and Middelburg, J.J. 2023. Aragonite dissolution protects calcite at the seafloor. Nature Communications 13: 1104
- ↑ Doo, S.S., Kealoha, A., Andersson, A., Cohen, A.L., Hicks, T.L., Johnson, Z.I., Long, M.H., McElhany, P., Mollica, N., Shamberger, K.E.F., Silbiger, N.J., Takeshita, Y. and Busch, D.S. 2020. The challenges of detecting and attributing ocean acidification impacts on marine ecosystems. ICES Journal of Marine Science 77(7-8): 2411–2422
- ↑ 17.0 17.1 Agostini, S., Harvey, B.P., Wada, S., Kon, K., Milazzo, M., Inaba, K. and Hall-Spencer, J.M. 2018. Ocean Acidification Drives Community Shifts towards Simplified Non-Calcified Habitats in a Subtropical-Temperate Transition Zone. Sci. Rep. 8, 11354
- ↑ Harvey, B.P., Kon, K., Agostini, S., Wada, S. and Hall-Spencer, J.M. 2021. Ocean Acidification Locks Algal Communities in a Species-Poor Early Successional Stage. Glob. Chang. Biol. 27: 2174–2187
- ↑ Somma, E., Terlizzi, A., Costantini, M., Madeira, M. and Zupo, V. 2023. Global Changes Alter the Successions of Early Colonizers of Benthic Surfaces. J. Mar. Sci. Eng. 11, 1232
- ↑ Czaja, R., Pales-Espinosa, E., Cerrato, R.M., Lwiza, K. and Allam, B. 2023. Using meta-analysis to explore the roles of global upwelling exposure and experimental design in bivalve responses to low pH. Science of the Total Environment 902, 165900
- ↑ Mele, I., McGill, R.A.R., Thompson, J., Fennell, J. and Fitzer, S. 2023. Ocean acidification, warming and feeding impacts on biomineralization pathways and shell material properties of Magallana gigas and Mytilus spp. Marine Environmental Research 186, 105925
- ↑ Vlaminck, E., Moens, T., Vanaverbeke, J. and Van Colen, C. 2022. Physiological response to seawater pH of the bivalve Abra alba, a benthic ecosystem engineer, is modulated by low pH. Marine Environmental Research 179, 105704
- ↑ Cordova-Rodríguez, K., Flye-Sainte-Marie, J., Fernandez, E., Graco, M., Rozas, A. and Aguirre-Velarde, A. 2022. Effect of low pH on growth and shell mechanical properties of the Peruvian scallop Argopecten purpuratus (Lamarck, 1819). Marine Environmental Research 177, 105639
- ↑ Steinmetz, J. C. 1994. Sedimentation of coccolithophores. In A. Winter, & W. G. Siesser (Eds.), Coccolithophores, (pp. 179–198). Cambridge University Press
- ↑ 25.0 25.1 Ziveri, P., Gray, W.R., Anglada-Ortiz, G., Manno, C., Grelaud, M., Incarbona, A., Rae, J.W.B., Subhas, A.V., Pallacks, S., White, A., Adkins, J.F. and Berelson, W. 2023. Pelagic calcium carbonate production and shallow dissolution in the North Pacific Ocean. Nature Communications 14: 805
- ↑ Krumhardt, K. M., Lovenduski, N. S., Long, M. C., Levy, M., Lindsay, K., Moore, J. K. and Nissen, C. 2019. Coccolithophore growth and calcification in an acidified ocean: Insights from community earth system model simulations. Journal of Advances in Modeling Earth Systems 11: 418-1437
- ↑ Marine Biogeochemical Library (MARBL), https://marbl-ecosys.github.io/
- ↑ Stockdale, A., Tipping, E., Lofts, S. and Mortimer, R.J.G. 2016. Effect of ocean acidification on organic and inorganic speciation of trace metals. Environ. Sci. Technol. 50: 1906–1913
- ↑ Millero, F.J., Woosley, R., Ditrolio, B. and Waters, J. 2009. Effect of ocean acidification on the speciation of metals in seawater. Oceanography 22: 72–85
- ↑ Whitby, H., Park, J., Shaked, Y., Boiteau, R.M., Buck, K.N. and Bundy, R.M. 2024. New insights into the organic complexation of bioactive trace metals in the global ocean from the GEOTRACES era. Oceanography 37: 142-155
- ↑ Cheriyan, E., Kumar, B.S.K., Gupta, G.V.M. and Bhaskara Rao, D. 2024. Implications of ocean acidification on micronutrient elements-iron, copper and zinc, and their primary biological impacts: A review. Marine Pollution Bulletin 199, 115991
Please note that others may also have edited the contents of this article.
|




