Difference between revisions of "Chlorpyrifos"
(ref) |
(→Notes) |
||
| Line 19: | Line 19: | ||
Chlorpyrifos enters the environment through direct application to crops, lawns and domesticated animals. They may also enter the environment through spills and the disposal of chlorpyrifos waste. | Chlorpyrifos enters the environment through direct application to crops, lawns and domesticated animals. They may also enter the environment through spills and the disposal of chlorpyrifos waste. | ||
| − | It has a low water solubility (0.7 mg/l) and a high tendency to [[adsorption|adsorb]] to particles and soils. Therefore it's unlikely to end up in the marine ecosystem. It has a high volatility causing it usually | + | It has a low water solubility (0.7 mg/l) and a high tendency to [[adsorption|adsorb]] to particles and soils. Therefore it's unlikely to end up in the marine ecosystem. It has a high volatility causing it after use to usually evaporate into the atmosphere , where it is degraded rapidly. It, however, only evaporates slowly from water bodies, mainly because chlorpyrifos in water is associated to suspended particles. In water it is slowly hydrolysed with a [[half-life]] of less than 60 days. <ref name="US">[http://www.atsdr.cdc.gov/toxprofiles/tp84.pdf U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES 1997 TOXICOLOGICAL PROFILE FOR CHLORPYRIFOS]</ref> |
| − | It has a high potential towards [[bioaccumulation]] and | + | It has a high potential towards [[bioaccumulation]] and might possibly also [[biomagnification|biomagnify]] through [[food chain|food chains]]. <ref name="US">[http://www.atsdr.cdc.gov/toxprofiles/tp84.pdf U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES 1997 TOXICOLOGICAL PROFILE FOR CHLORPYRIFOS]</ref> |
Chlorpyrifos is very toxic for crustaceans, especially amphipods which die at concentrations above 0.1 µg/l. Some fish species start dying at concentrations above 1 µg/l while other can tolerate concentrations above 500 µg/l. <ref>[http://www.pesticideinfo.org/List_AquireAcuteSum.jsp?Rec_Id=PC33392 www.pesticideinfo.org]</ref> | Chlorpyrifos is very toxic for crustaceans, especially amphipods which die at concentrations above 0.1 µg/l. Some fish species start dying at concentrations above 1 µg/l while other can tolerate concentrations above 500 µg/l. <ref>[http://www.pesticideinfo.org/List_AquireAcuteSum.jsp?Rec_Id=PC33392 www.pesticideinfo.org]</ref> | ||
Revision as of 11:38, 27 August 2009
Definition of Chlorpyrifos:
Chlorpyrifos is an insecticide that has been widely used to control cockroaches, fleas, termites and to control crop pests. In 1997, chlorpyrifos was withdrawn from most indoor and pet uses. Chlorpyrifos is a white crystal-like solid with a strong odour. [1]
This is the common definition for Chlorpyrifos, other definitions can be discussed in the article
|
Notes
| Chlorpyrifos |
|---|
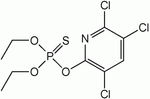
|
| Formula |
| C9H11Cl13NO3PS |
Chlorpyrifos enters the environment through direct application to crops, lawns and domesticated animals. They may also enter the environment through spills and the disposal of chlorpyrifos waste.
It has a low water solubility (0.7 mg/l) and a high tendency to adsorb to particles and soils. Therefore it's unlikely to end up in the marine ecosystem. It has a high volatility causing it after use to usually evaporate into the atmosphere , where it is degraded rapidly. It, however, only evaporates slowly from water bodies, mainly because chlorpyrifos in water is associated to suspended particles. In water it is slowly hydrolysed with a half-life of less than 60 days. [1]
It has a high potential towards bioaccumulation and might possibly also biomagnify through food chains. [1]
Chlorpyrifos is very toxic for crustaceans, especially amphipods which die at concentrations above 0.1 µg/l. Some fish species start dying at concentrations above 1 µg/l while other can tolerate concentrations above 500 µg/l. [2] In humans, chlorpyrifos can cause cholinesterase inhibition; it can overstimulate the nervous system causing nausea, dizziness, confusion, and at very high exposures respiratory paralysis and death. [3]
Environmental standards and legislation
Included in the water framework list of priority substances
See also
Chlorpyrifos on the ED North Database